With the dust yet to settle on the US FDAs inspection of Sun Pharmaceuticals Mohali, Punjab and Halol, Gujarat plants, 2 more companies have reported receiving Form 483 for their non compliance to cGMP – Current Good Manufacturing Practices.
Related Post: Tracking The Sun Pharma Halol Plant US FDA Inspection

Divis Laboratories Unit-II at Visakhapatnam, Andhra Pradesh was inspected by the US FDA team from the 29-Nov to 6-Dec. While the detailed observations have not been disclosed, Divis has confirmed that there were 5 observations made by the US FDA team.
This second facility is an Export Oriented Unit and has 8 production blocks. It is situated on a 100-acre site which went into commercial operations since March, 2003.

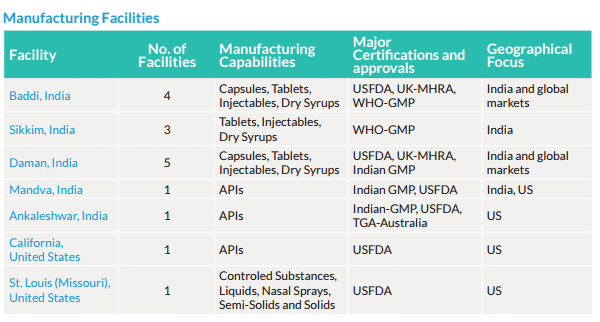
Alkem Laboratories API (Active Pharmaceutical Ingredient) manufacturing facility located at Ankleshwar, Gujarat was inspected by the US FDA team from the 5-Dec to 9-Dec. While the detailed observations have not been disclosed, Alkem has confirmed that there were 3 observations made by the US FDA team.
Just 2 months ago in Sep-16, US FDA inspected Alkem’s Daman facility and issued 13 observations and surprisingly both facilities – Daman and Ankleshwar were actually inspected just last year.
Alkem has filed over 60% of ANDA from Daman and over 30% from the Ankleshwar site.
Both the companies are now putting together a detailed response with adequate corrective and preventive measures to address the US FDA Observations.
Since the Form 483 for these inspections have not been made public yet, we will closely watch for any updates from the companies themselves and the FDA and keep you updated.
Alkem Laboratories & Divis Laboratories Receive Form 483 From US FDA

Like our content? Join Capitalmind Premium.
- Equity, fixed income, macro and personal finance research
- Model equity and fixed-income portfolios
- Exclusive apps, tutorials, and member community
Subscribe Now
Or start with a free-trial
Already a subscriber?
Login Now




